Along with air, Water is essential for the existence of mankind. Water exists above the earth’s surface [in the form of clouds, fog, dew] and also below the earth surface.
Water is essential for the sustainability of human life. Water is a compound of hydrogen and oxygen in the ratio of 2:1.
Occurrence of Water
Water can be found in different parts of the earth. They are:
Oceans

They include the oceans, seas, rivers and lakes. They account for 3/4th of the water content on planet earth.
- Seawater has dissolved impurities.
- Dissolved impurities include unwanted minerals, salts and dissolved gases.
- River water has suspended impurities.
- Suspended impurities include clay, mud, gravel, dead plants and animals.
Clouds and Rain

The water from cloud and rain is derived from the water cycle itself.
The water which is available from the rain contains dissolved gases like oxygen and carbon dioxide in the first showers.
Glaciers and Snow
- They account for 2% of the total water available on earth.
- But most importantly they supply 98% of the total fresh water on earth.
- So, Glaciers are essential for the sustainability of life on Earth.
More facts about the how important water is to sustain life on earth:
- The human body has a water content of 70%. And a loss of even 2% of this water can be harmful to life and leads to complete memory loss.
- The fruits and vegetables which are an important part of any diet has a water content of 75-80% which makes the water very essential for living beings on earth
Water Cycle
Water cycle can be defined as the circulation of water from the ‘earth surface’ to the atmosphere and then back to the earth again.
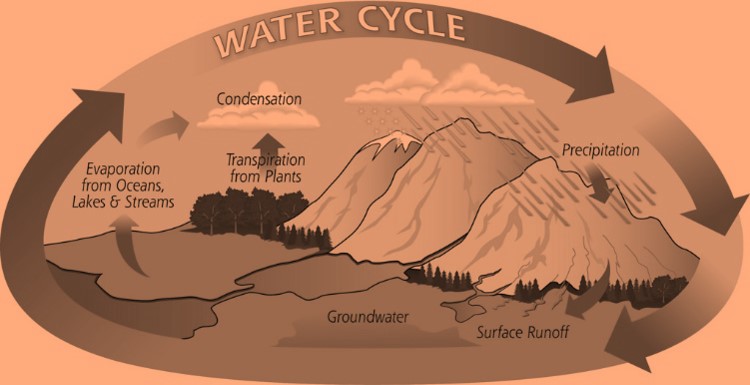
This water cycle is very important for life on earth and this maintains the essential balance required for earth to be full of life.
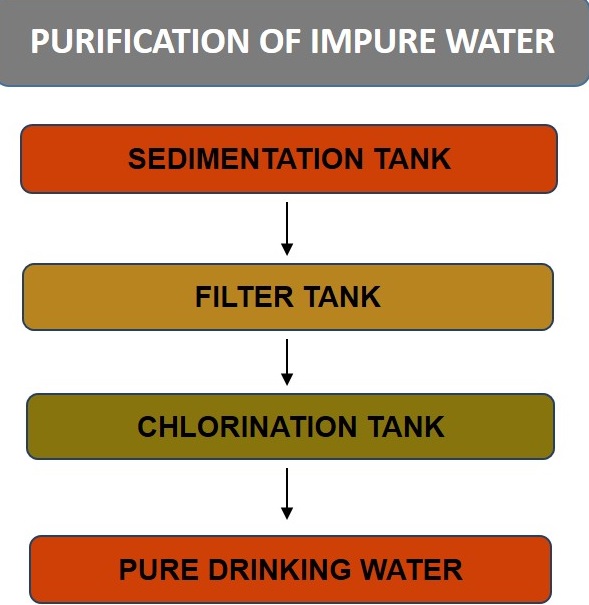
Purification of Impure Water
Purification of natural water for drinking purpose is generally done by passing the water through three tanks. They are:
Purification of sea water is done by- Distillation process
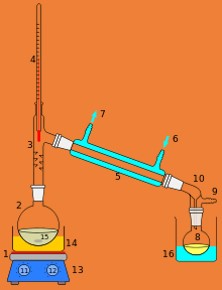
- Impure sea water is added to the flask of the apparatus.
- On heating the flask, the pure water evaporates and the vapours condense by the passage through the condenser.
- The condensed water is received in the smaller flask.
Properties of Water
- Colour: Water is a colourless liquid.
- Odour: Water is an odourless liquid.
- State: Water can exist in all three states: Ice(Solid), Water(Liquid), Water vapour(Gas).
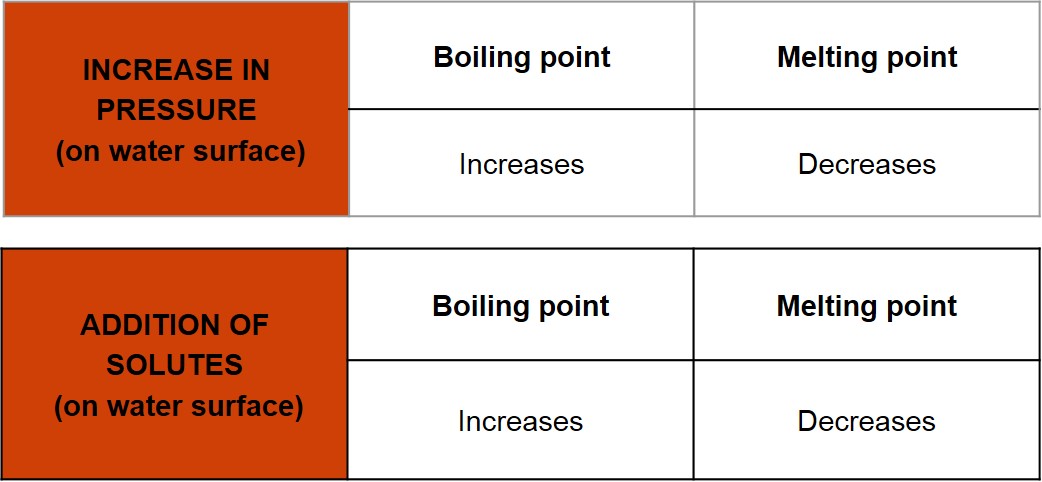
- Boiling Point: 100 degree Celsius at 760mm pressure.
- Melting Point: 0 degree Celsius at 760mm pressure.
Anomalous Expansion of Water
- Al liquid contracts on cooling.
- Water also contracts on cooling till it reaches a temperature of 4 Degree Celsius.
- On Cooling below 4 Degree Celsius up to Melting Point, water expands instead of contracting.
- This behaviour of water is known as “anomalous".
- The density of water is highest at 4 Degree Celsius.
Examples: Ice floats on the surface of water and forms an insulating layer on the water allowing survival of fishes and aquatic animals below it.
This is one of the main applications of ‘anomalous expansion of water’.
The Conductivity of Water
- Thermal: Water is a bad conductor of heat.
- Electricity: Water is a bad conductor of electricity.
Tests for Water
Physical tests
The boiling and freezing points of water are taken as the physical tests.
- Water is taken in a test tube
- It is heated or put on crushed ice.
- The water is heated until it forms steam or cooled until it forms ice.
- The constant Temperature is noted.
- Thus, we become sure that it is water by seeing the temperatures
Chemical Tests
Water turns Anhydrous Copper Sulphate Blue is the chemical test.
Solubility
- Solubility of most solids in water increase with an increase in temperature.
- Liquids which are soluble in water is called miscible liquid.
- Solubility of gases is inversely proportional to the temperature and directly proportional to the pressure of water.
Recap
- Water is essential for the sustainability of human life. Water is a compound of hydrogen and oxygen in the ratio of 2:1.
- Water is found naturally in oceans, clouds and glaciers.
- Water cycle can be defined as the circulation of water from the ‘earth surface’ to the atmosphere and then back to the earth again.
- Purification of sea water is done by the distillation process.
- Ice floats on the surface of the water and forms an insulating layer on the water allowing survival of fishes and aquatic animals below it.
- The boiling and freezing points of water are taken as the physical tests.
- Water turns Anhydrous Copper Sulphate Blue is the chemical test.
- Water is a bad conductor of heat and electricity.
























Comments