Substances can be divided into three types –
- Acids
- Bases
- Salts
Acid
Acid tastes sour. The substances that contain acid and are sour are lemon, curd, orange juice, pickles, vinegar, etc.
These substances are called as Acidic. The word acid is derived from the Latin word “ACERE" that means sour.
Base
Base tastes bitter. Substances that have bitter taste are soap solution, baking soda, soap, washing soda, etc.
The substances that contain base and have chemical nature are known as BASIC.
Characteristics of Acid
- Acid is sour in taste.
- It converts blue litmus paper into the red.
- It also converts China rose solution into dark pink color.
Characteristics of Base
- A base is bitter in taste.
- It converts red litmus paper into blue color.
- It turns china rose solution into green color
Indicator
Understanding whether the nature of substances is acidic or basic cannot always be understood by tasting them. Tasting in a laboratory is also harmful thus for this purpose a special substance is used called an indicator.
It is a substance that tells about the acidic or basic nature of a particle only by changing the color.
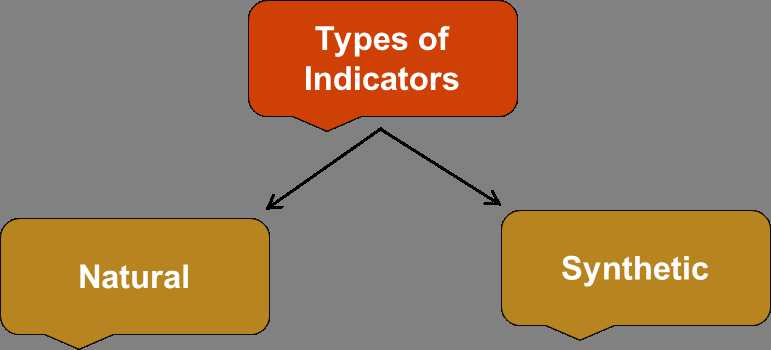
- Natural Indicator – When the indicators are obtained from the naturally occurring substances than they are known as Natural Indicators. For example – China rose, litmus, turmeric, etc.
- Synthetic Indicator – When the indicators are made in the laboratory then they are called as synthetic indicators. For example – methyl orange, phenolphthalein, etc.
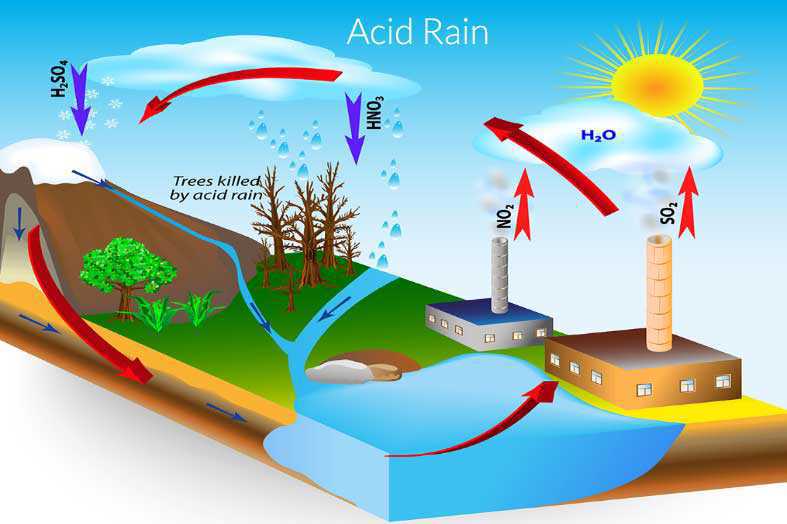
Acid rain
When carbon dioxide, nitrogen dioxide and sulphur dioxide are released from vehicles then mix with the rain droplets and turn it too acidic. The rainwater becomes acidic and when the rain falls it is called as acid rain. Acid rain is harmful to buildings and plants and animals. Acid rain has also affected Taj Mahal.
Neutralization
When solutions of acid and base are mixed then they both neutralize each other and form a third substance called SALT. This process is known as neutralization. The solution of salt is neither acidic nor basic and hence called as a neutral solution.
When a base called sodium hydroxide is mixed with hydrochloric acid then they form a common salt called as sodium chloride.
Sodium Hydroxide + Hydrochloric acid Sodium chloride + Water
(Solution Solution Solution Liquid)
Neutralization in Soil Treatment
Due to the more use of fertilizers or incorrect use of harvesting methods, the soil becomes acidic or basic. It affects the growth of plants. Then quicklime and slaked lime are used with Acidic soil to nullify the acidic nature of the soil. In case of basic soil, it is combined with organic matter.

Recap
- Acids are sour in taste and bases are bitter.
- There are three types of substances-
- Acid
- Base
- Salt
- Salts are formed when with the combination of Acids and Bases. This process is called as Neutralization.
- The two types of Indicators are –
- Natural Indicators
- Synthetic Indicators































Comments