When energy is transferred from a hot body than it is called as heat. The degree of hotness or coldness of something can be understood by the sense of touch. But this sense of touch is risky and cannot be reliable always. Hence temperature is used to measure the hotness of anything in a reliable manner. The device used to measure temperature is called as Thermometer.
Unit of Heat
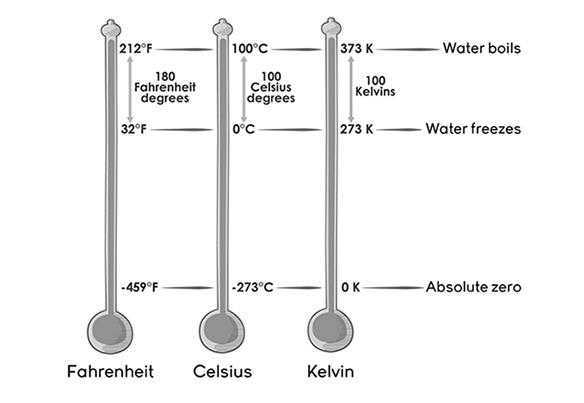
Heat or temperature is measured in three units-

- Celsius – It is written as °C and read as degree Celsius. For example; 20°C is read as twenty degree Celsius.
- Fahrenheit – It is written as °F and read as degree Fahrenheit. For example; 35°F is read as Thirty-five degree Fahrenheit.
- Kelvin – It is written as K. For example; 50K is fifty Kelvin.
Thermometer
The device used to measure temperature is known as Thermometer. It has a bulb at one end and is made up of a long narrow glass tube. Since the tube is filled with mercury hence it appears as a continuous silver line. At room temperature, mercury is in liquid state and contracts or expands with the change in temperature.
Types of Thermometer
1. Laboratory Thermometer
The scale of laboratory thermometer generally ranges from -10°C to 110°C over the glass tube. Further, there is a division of temperature scale into 10 parts to read the fraction of temperature.

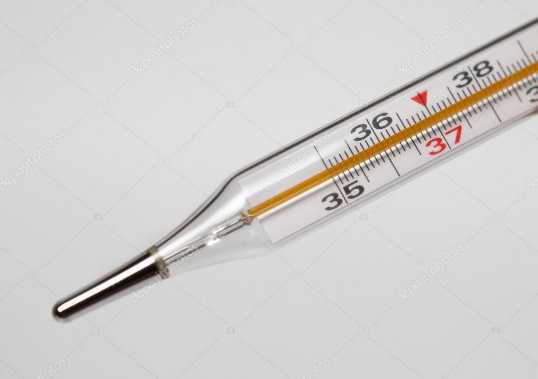
2. Clinical Thermometer
It is used to measure the body temperature. The scale of clinical thermometer generally ranges from 35°C to 42°C and or from 94°F to 108°F because human body temperature remains in this range only. Near the bulb of the clinical thermometer, there is a kink made to prevent the automatic fall of mercury level.
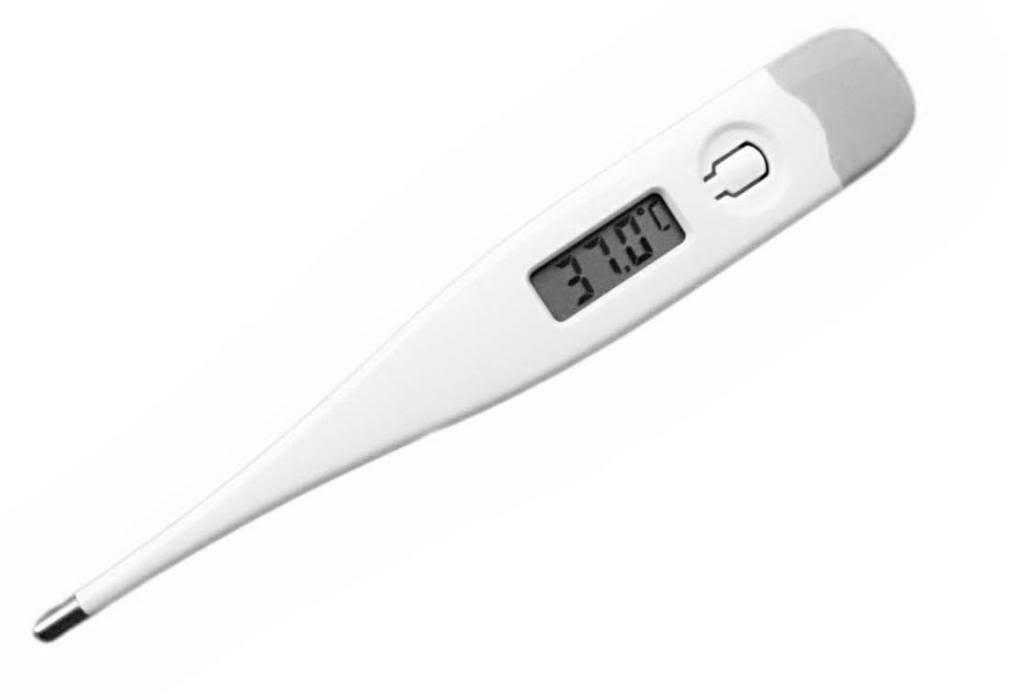
3. Digital Thermometer
Just like digital watches, in a digital thermometer, the reading of temperature is displayed digitally. Due to no use of mercury, this is safe.
4. Maximum-Minimum Thermometer
The daily temperature required to prepare the weather reports come from this thermometer.

Transfer of Heat
Heat takes place when the temperature an object is different from its surroundings. Heat brings the body and surroundings at the same temperature. The transfer of heat takes place from hotter to cooler objects. Heat is transferred by conduction, convection and radiation.
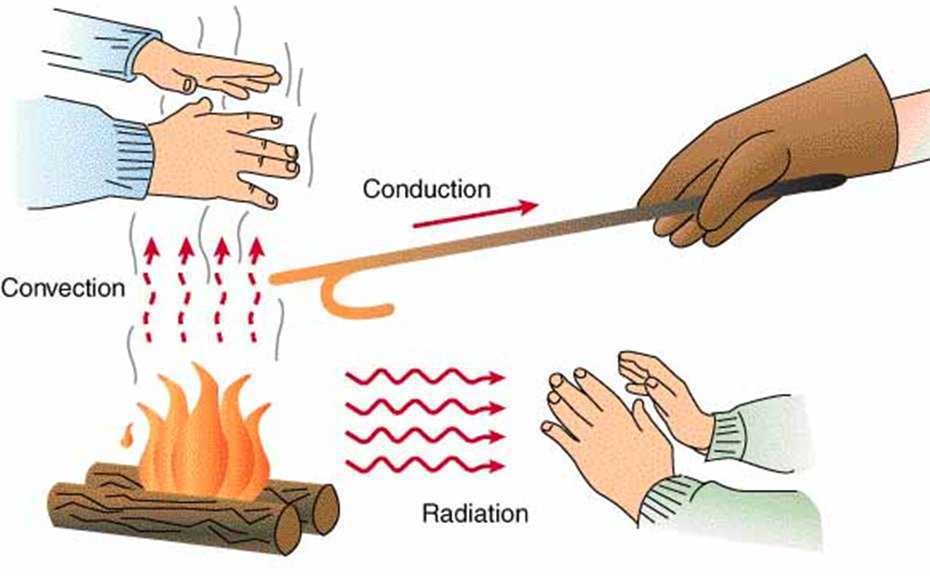
1. Conduction
When heat is transferred from one particle to the adjacent particle than it is called as conduction of heat. The process of transferring heat in solids takes place through conduction. This process takes place through adjacent molecules. The materials which allow the heat to pass through it are called as conductors like copper, iron, etc.
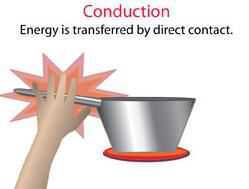
For example – When you put one end of the rod over the flame than that particular part gets heated first and then slowly heat gets transferred to the other end of the rod. This happens because the iron particle which is nearer to the flame gets heat and transfers to the adjacent particles. Thus it proves that heat transfer takes place through conduction in solids.
2. Convection
When the transfer of heat takes place due to the movement of molecules of a medium then it is called as convection of heat. Convection takes place only in liquids and gases.

For example – In a building lower floor is cooler than the upper floor, wind currents, water at the surface of a lake or swimming pool is cooler.
3. Radiation
When the heat flows from one source to another through medium or vacuum than this process is called as radiation. When the surroundings of the body absorb heat than the temperature increases.

For example – Microwave oven, a campfire, light bulb, etc.
Recap
- When a hot body releases the energy than it is known as Heat.
- Unit of heat is measured in three units – Celsius, Fahrenheit & Kelvin
- The device used to measure temperature is called as the thermometer. It has a bulb and a glass tube attached to it.
- There are four types of the thermometer- Laboratory thermometer, Clinical thermometer, Digital thermometer & Maximum – minimum thermometer
- Transfer of heat takes place from one object to another. There are three methods to transfer heat Conduction, Convection & Radiation































Comments